A Close Encounter With The Bionic Pancreas
I’m now exactly eleven days out of the five-day Boston University/Massachusetts General Hospital bionic pancreas trial that I took part in. I can’t quite talk about it too directly, without my eyes starting to fill up. So dramatic, I know! But I feel forever changed, having been part of this research study, and now knowing (albeit only for a week) what “normal” life was like before diabetes – not having to carb count, not experiencing any hypoglycemia, not feeling tired all the time, and not having diabetes on my mind every five minutes. In short, I felt like a whole new Kelly. You can see some of the pictures and videos I took on our diaTribe twitter at twitter.com/diaTribenews.
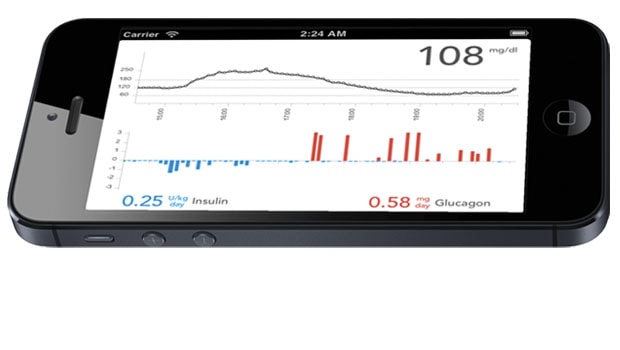
What is this study exactly? Well, Dr. Edward Damiano (the engineering genius from BU), Dr. Steven Russell (a star endocrinologist at MGH), and a slew of colleagues have built a system over the last decade to make life with type 1 diabetes much, much easier. The system uses an iPhone running a control algorithm, two Tandem t:slim insulin pumps (dosing insulin and glucagon), and a Dexcom G4 continuous glucose monitor. This bionic pancreas (also called an “artificial pancreas” or “closed loop”) takes glucose data from the Dexcom CGM and runs it through a control algorithm on the iPhone. The algorithm then processes the data and automatically directs insulin and glucagon dosing – in a word, magical. This dosing happens every five minutes, so the system automatically makes many very small adjustments to keep glucose levels in range. The bionic pancreas only needs your weight to start up, and the algorithm adapts and learns over time from its performance – in other words, if I become very insulin resistant for a few days (e.g., little sleep, stress, less than my normal amount of exercise), it will robustly adapt and increase the amount of insulin it gives. How cool is that!
What was also so impressive about Drs. Damiano and Russell’s study was how very real-world it was. Unlike most studies in the past that confined bionic pancreas research participants to a hospital bed, I was able to roam around a three-square mile area of Boston. I could eat what I wanted (restaurants were encouraged!), exercise as I wanted, and sleep by myself in a hotel room. Just in case anyone was ever close to danger, the study had three teams of nurses following me and making sure I stayed safe, even to the point where I wore an IV at night and they followed my minute-to-minute blood glucose readings on a screen next door. Big picture, it was fantastic to see what wearing a bionic pancreas would be like in more of a daily life setting. The study’s goal is to assess the safety and effectiveness of the bionic pancreas, so its performance was compared to five days of my own “standard” care without the bionic pancreas.
![]() I knew it would be cool to be part of the trial. I’m one of about 30 participants who get to take part in it, one of the first closed-loop trials in the world to take place outside the hospital setting. I love new technology, I love clinical trials, and I love the hope that accompanies early-stage research.
I knew it would be cool to be part of the trial. I’m one of about 30 participants who get to take part in it, one of the first closed-loop trials in the world to take place outside the hospital setting. I love new technology, I love clinical trials, and I love the hope that accompanies early-stage research.
So when they told me that I might be quite depressed when I had to give back the bionic pancreas (a little box that the research team kept calling “your pancreas,” as in “Oh, don’t forget your pancreas!”, “Don’t drop your pancreas…”, etc.) I felt “Oh right! Come on. This is so exciting being part of this trial. How could I ever be depressed!”
How, indeed.
So what was it like to have a machine making all these decisions for me? Well…
• I was never hypoglycemic
• I never felt hypoglycemic
• I was never worrying about hypoglycemia
• I was never recovering from hypoglycemia
That’s already enough of a game changer for me. But there was more…
• If I started veering low, my bionic pancreas figured it out and gave me the perfect amount of glucagon to make sure that hypoglycemia didn’t occur
• I always felt safe during the week – at no time did I feel threatened or scared
• My glucoses were being watched and stayed perfectly in range overnight, every night. Wow.
• I counted zero carbs
• I never “corrected”
• I never thought about insulin sensitivity and how I couldn’t figure that out
• I never thought about insulin to carb ratios
• I never bolused
• I was a nicer and kinder person the entire week with the bionic pancreas
Because the system uses both insulin and glucagon, I got to see mini doses of glucagon in action for the first time. There’s no question about it – glucagon is a cool hormone. It is ten times better than orange juice, glucose tabs, candy, gel, etc. It is SO precise and the power of having just the right amount was really cool. It was magic!
It is SO precise and the power of having just the right amount was really cool. It was magic!
Don’t get me wrong – the system still doesn’t match how great a pancreas is in someone without diabetes. The bionic pancreas must still deal with the 60-90 minute delays in rapid-acting insulin absorption. That means that my blood sugar did exceed 200 mg/dl after big meals. (An analogy to think about this is steering a car – if it took the car 60-90 minutes to respond to movements of the steering wheel, you would inevitably run off the road sometimes.) What was very key was how the system brought me back down from these highs – super safely and far more quickly than I could regularly ever do. I went from a high over 200 mg/dl to a safe, soft landing right in range. Usually when I stare at a number over 200 mg/dl, I take too much insulin (a “rage bolus” as Kerri Sparling would say), go low, eat too much, go high, and ride a roller coaster of highs and low all day.
During the trial, I also had a striking epiphany about living with diabetes: “Oh my gosh, I waste so much time having diabetes!” Being distracted because of a low, doing all these things to make sure I’m staying in range, and the super big time-leech, hyperglycemia. Whew! I felt like my whole world changed when I was constantly in a state of normoglcyemia. And then I wondered how much I try to be “normal” and make it “seem” like diabetes is easy to manage – that mentality is also probably exhausting, even though I’m not even aware of it.
Getting rid of hypoglycemia and hyperglycemia for a week was one of the most powerful things I’ve ever experienced.
My bionic pancreas never failed me. I felt like the 17 year-old, completely carefree Kelly I used to know back before I was diagnosed. Getting rid of hypoglycemia and hyperglycemia for a week was one of the most powerful things I’ve ever experienced. I already like myself but with the bionic pancreas, I felt even better in my own two shoes. And in turn – this experience gave me renewed hope for what I used to say might be possible and what I know today absolutely is possible.
![]() The fine print: There are a lot of things that have to go well in order for this to be commercially available by 2017. That is the year that Dr. Damiano’s son will go to college and the year that Dr. Damiano hopes to see a commercial system approved by the FDA. Here’s what has to happen:
The fine print: There are a lot of things that have to go well in order for this to be commercially available by 2017. That is the year that Dr. Damiano’s son will go to college and the year that Dr. Damiano hopes to see a commercial system approved by the FDA. Here’s what has to happen:
•The rest of the current trial has to go well so that the researchers can move onto the next phase – testing the system at diabetes camps this summer (they will be followed by one nurse per four kids).
•After that, Drs. Damiano and Russell’s team will test the bionic pancreas in healthcare providers who have diabetes themselves. The two-week study will be more ambitious, as the participants will sleep in their own homes and go to work as they normally would. (This is pretty smart right – they still have healthcare providers around, to ensure safety, but they are themselves!)
•If all goes well, the research group plans to conduct pivotal studies, at least one of which will last six-months, that will test the final version of the system in 2015 and 2016.
•For these longer-term trials, a new pump will need to be built and approved that can carry both insulin and glucagon. Tandem Diabetes Care is currently working on developing such a pump (see our new now next from diaTribe #50 on this news).
•A stabilized liquid glucagon must be developed that can last for a few days in a pump. In this study, the researchers used the current glucagon “kit,” which requires mixing glucagon powder with water. Unfortunately, it had to be replaced every day in the glucagon t:slim pump I was wearing. While this was acceptable in a research setting, it’s not a viable real-world solution because it is too expensive (it’s also a pain, though I would do it in a heartbeat). We believe that this is the biggest obstacle to overcome in the next few years. Fortunately, companies such as Xeris Pharmaceuticals, Biodel, and others are working hard on addressing this problem. The timeline will be tight to make the goal, though I’m optimistic.
•Before it can be sold, the FDA has to approve the bionic pancreas – certainly no easy task considering the ongoing three-plus-year delay in approving Medtronic’s MiniMed 530G. Known as the Veo outside the US, this is the most basic version of an automated insulin delivery system – it suspends insulin delivery for up to two hours when the CGM crosses a low threshold and the user does not respond to an alarm. We understand that Dr. Damiano and colleagues’ interactions with the FDA have been quite constructive, so we are hopeful here as well, though it may be a complex journey. Indeed, given last year’s artificial pancreas guidance from the FDA, there does seem to be a clear approval path forward. (For more on that topic, see the learning curve in diaTribe #39.)
I had been warned that I wouldn’t want to give my bionic pancreas back to the researchers at the end of my five days with it. They said I might become depressed. I laughed – who could possibly become depressed after being so lucky to be chosen for this trial?
Well, they were right – I definitely did not want to give it back. And I certainly miss it.