Type 2 Insulin Therapy: Wherever You Go, V-Go

Among all the nations where diabetes is a serious health concern, the United States is an outlier when it comes to insulin treatment therapies. While insulin pens rule the roost among multiple daily injectors in Japan, and in many European countries, the good old-fashioned syringe is still preferred by a majority of insulin users here. This is particularly true for Type 2s using insulin, for whom full insurance reimbursement is often guaranteed only for the least expensive forms of insulin delivery. For Type 2s, insulin pumps, and the more effective therapy they have been shown to provide, can be difficult to acquire. In part, this is due to the high burdens of proof that many insurers, Medicare among them, still require to switch a patient from injection to infusion. Essentially, all other control therapies must have failed. If payors resist, and many primary care doctors continue to see insulin as a last resort rather than an initial therapy, most patients’ natural aversion to shots or invasive infusion sets will combine to create a triple whammy of issues that damage effective diabetes management among a large and growing number of Type 2 patients who need insulin.
Although most Type 2s still produce some insulin naturally, many develop autoimmune characteristics as they age (sometimes they are labeled “Type 1.5” by experts), and require man-made insulin to keep their glucose levels in check. Even though Type 2s may use far more insulin than Type 1s (a function of their insulin resistance), the daily routine for people who have no functioning beta cells is far more complex than that needed by most Type 2 patients. Traditional insulin pumps are massively overbuilt for the needs of many Type 2s, because they are designed to treat people whose pancreas no longer produces any insulin.
What this market needs is a good, inexpensive solution to insulin delivery, one that doesn’t require shots or a full-blown insulin pump. Enter Valeritas (www.valeritas.com), a medical device company headquartered in Bridgewater, NJ. The company has stepped into this treatment gap with V-Go, an insulin delivery system specifically designed for the needs of Type 2s (or Type 1.5s). V-Go made its debut in 2012. It is alluringly simple to use, easy to control, and best of all, inexpensive compared to other pump brands.
It’s Not A Patch
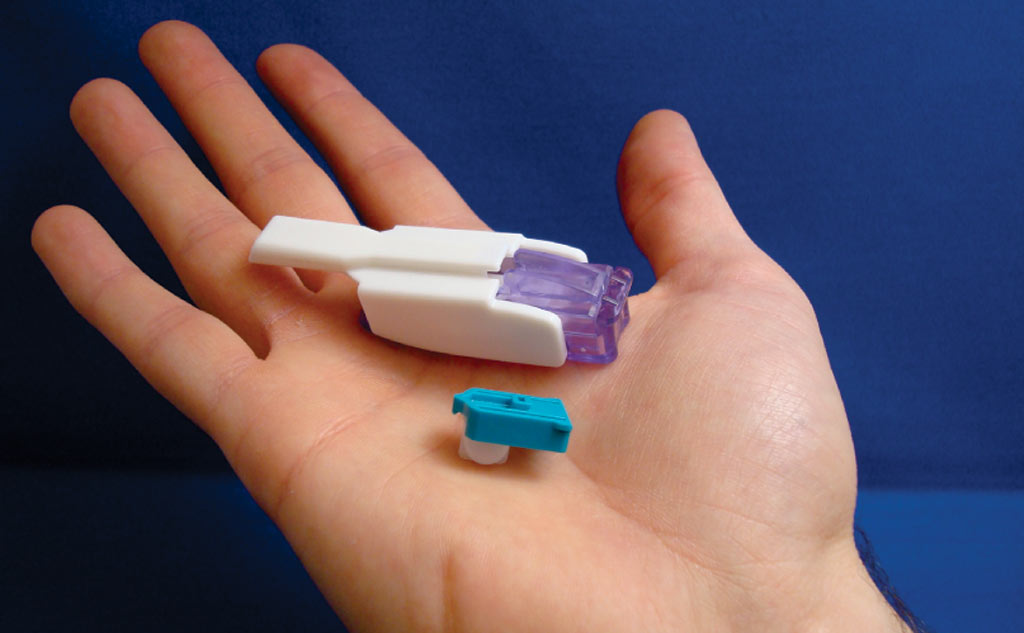
 Despite its patch-like appearance and adhesive connection to the body, V-Go isn’t a patch pump. Because it delivers insulin through a tiny needle that provides subcutaneous infusion, V-Go is rated as a device in the same class as a syringe, something that should ultimately please both insurers and patients. Valeritas created this new insulin device — which it stresses, is neither pump nor pen — to dispense insulin for 24 hours, and then be discarded. While it’s attached to the body, V-Go offers continuous hourly insulin doses for 24 hours, along with on-demand mealtime boluses. There are no electronics, batteries, infusion sets, or programming. It’s been tested in water to depths of 3 feet, 3 inches (1 meter) for 24 hours, so it can be used in the shower or even when swimming.
Despite its patch-like appearance and adhesive connection to the body, V-Go isn’t a patch pump. Because it delivers insulin through a tiny needle that provides subcutaneous infusion, V-Go is rated as a device in the same class as a syringe, something that should ultimately please both insurers and patients. Valeritas created this new insulin device — which it stresses, is neither pump nor pen — to dispense insulin for 24 hours, and then be discarded. While it’s attached to the body, V-Go offers continuous hourly insulin doses for 24 hours, along with on-demand mealtime boluses. There are no electronics, batteries, infusion sets, or programming. It’s been tested in water to depths of 3 feet, 3 inches (1 meter) for 24 hours, so it can be used in the shower or even when swimming.
V-Go uses rapid-onset insulin, either Humalog of Novolog, which the user loads into the device. About 2.5 inches by 1 inch, and about as thick as an OmniPod pod, V-Go is placed on the skin, and begins to dose insulin when the small needle on the skin-facing side of the device is inserted into the skin. V-Go is inexpensive to buy (less than $300 over the counter if no insurance is available) because it’s inexpensive to make. Essentially the device is built around principles of fluid dynamics, using pressure from an uncoiling spring inside the V-Go to gradually push insulin through the tip of the device. Look, ma – no circuit boards!
There are 3 options a doctor can prescribe: V-Go 20, V-Go 30, and V-Go 40. The numbers refer to insulin units on board for hourly basal infusion rates of .83 units, 1.25 units, or 1.67 units. All 3 models also hold 36 units of insulin that can be infused as bolus doses in 2-unit increments by pushing a button on the face of the device as many times as the bolus requires – 3 button-pushes for 6 units, 4 button-pushes for 8 units, and so on. You refill a new device, attach it in a different location at least 2 inches away from the previous site, and throw away the old device every 24 hours. Don’t want to reload insulin every day? Valeritas says you can load Hunalog 24 hours in advance, whether you refrigerate it or leave it at room temperature, and that Novolog can be loaded and stored up to 5 days before use if refrigerated, or 3 days in advance at room temperature.
Betting On a Sea Change
 Investors have backed Valeritas with $250 million in combined equity and debt financing, betting that the time is right for an insulin infusion platform that is simple and inexpensive enough to be used by millions of patients. Indeed, the Type 2 market dwarfs the Type 1 universe worldwide, but this really isn’t a Type1 vs. Type 2 contest. Instead, it’s a bet on a more widespread adoption of insulin therapy as an early intervention in Type 2 cases, rather than the traditional “when all else fails” practice that is still standard operating procedure for many primary care physicians. Educating the primary care marketplace, where most Type 2s get their care, is one of the major challenges facing Valeritas.
Investors have backed Valeritas with $250 million in combined equity and debt financing, betting that the time is right for an insulin infusion platform that is simple and inexpensive enough to be used by millions of patients. Indeed, the Type 2 market dwarfs the Type 1 universe worldwide, but this really isn’t a Type1 vs. Type 2 contest. Instead, it’s a bet on a more widespread adoption of insulin therapy as an early intervention in Type 2 cases, rather than the traditional “when all else fails” practice that is still standard operating procedure for many primary care physicians. Educating the primary care marketplace, where most Type 2s get their care, is one of the major challenges facing Valeritas.
Another potential obstacle is reimbursement by health insurers, always a hurdle for a new device. As mentioned above, this should not be a problem in the long run, due to the way the device is classified, but V-Go’s builders don’t expect their customers to go it alone. The company provides 24/7/365 telephone support through its call center (866-881-1209), working with potential patients to find out how much of V-Go’s costs will be covered by their insurance companies. The call center also offers support to healthcare providers. Valeritas currently employs about 75 full-time salespeople, whose efforts are focused on doctors. Is there a direct-to-consumer pitch in V-Go’s future? The company won’t say, but the available resources suggest one might be possible.
How Well Does V-Go Work?
 Naturally, the most important aspect of V-Go is its performance in improving glycemic control among users. Because the product’s preset hourly units combined with limited-option boluses dispense insulin therapy differently than traditional pumps, V-Go may have a different impact on patients than injections of long-acting insulin like Lantus combined with rapid-onset mealtime insulin. An ongoing observational study, that enrolled Type 2s with a baseline A1C greater than 7 percent, studied their treatment results using baseline (non-V-Go) therapy for 4 to 6 weeks before switching them to V-Go for both basal and mealtime insulin use. A recently released interim analysis of 47 men, who used V-Go continuously for 3 months after being observed using different insulin therapies, showed their A1C levels declined from an average 9.0 percent to 8.6 percent during the 4 to 6 week non-V-Go phase (there’s nothing like a clinical trial to make people pay attention), and a further improvement of A1C from 8.6 percent to 7.8 percent during the 3-month V-Go period. The greater percentage decrease in A1c levels with V-Go use supports an apparent improvement in therapy as a result of the platform. Five patients reported adverse events probably related to V-Go (primarily rash or skin irritation). Six patients reported hypoglycemia, with glucose levels below 70 mg/dL. There was no significant change in weight, and total daily insulin dose was reduced by 19 percent (12.5 units) on average.
Naturally, the most important aspect of V-Go is its performance in improving glycemic control among users. Because the product’s preset hourly units combined with limited-option boluses dispense insulin therapy differently than traditional pumps, V-Go may have a different impact on patients than injections of long-acting insulin like Lantus combined with rapid-onset mealtime insulin. An ongoing observational study, that enrolled Type 2s with a baseline A1C greater than 7 percent, studied their treatment results using baseline (non-V-Go) therapy for 4 to 6 weeks before switching them to V-Go for both basal and mealtime insulin use. A recently released interim analysis of 47 men, who used V-Go continuously for 3 months after being observed using different insulin therapies, showed their A1C levels declined from an average 9.0 percent to 8.6 percent during the 4 to 6 week non-V-Go phase (there’s nothing like a clinical trial to make people pay attention), and a further improvement of A1C from 8.6 percent to 7.8 percent during the 3-month V-Go period. The greater percentage decrease in A1c levels with V-Go use supports an apparent improvement in therapy as a result of the platform. Five patients reported adverse events probably related to V-Go (primarily rash or skin irritation). Six patients reported hypoglycemia, with glucose levels below 70 mg/dL. There was no significant change in weight, and total daily insulin dose was reduced by 19 percent (12.5 units) on average.
“This observational study enables us to obtain data about the actual clinical benefit of the V-Go when used as directed by practicing physicians in a real-world setting,” said Dr. Cheryl Rosenfeld, primary study investigator affiliated with North Jersey Endocrine Consultants. “These interim results are very promising, suggesting improved glycemic control, a reduction in total daily insulin dose with a nominal change in weight in patients switching to the V-Go.”
In other words, as is most often true, infusion therapy trumps injections as an effective way to control glucose while helping to reduce high and low excursions and decrease insulin use. While the current study is not complete, and more observational studies will no doubt be conducted, V-Go seems to be a safe and useful tool for improving glycemic control among Type 2 patients. The biggest remaining challenge to more widespread use is familiarizing prescribing doctors, particularly non-specialists, of the overall benefits of early insulin therapy.
Thanks for reading this Insulin Nation article. Want more Type 1 news? Subscribe here.
Have Type 2 diabetes or know someone who does? Try Type 2 Nation, our sister publication.