Afrezza Enters the Home Stretch…Again
On August 14th, MannKind Corporation executives briefed press and investment analysts on the positive results reported for two Phase 3 clinical trials of Afrezza, an inhalable insulin product in development for the past 12 years. (Read “Inhalable Insulin: A Breathtaking Development” at Insulin Nation). It’s another step forward in MannKind’s long trek through the regulatory wilderness, a journey which has included two previous FDA refusals to clear the product.
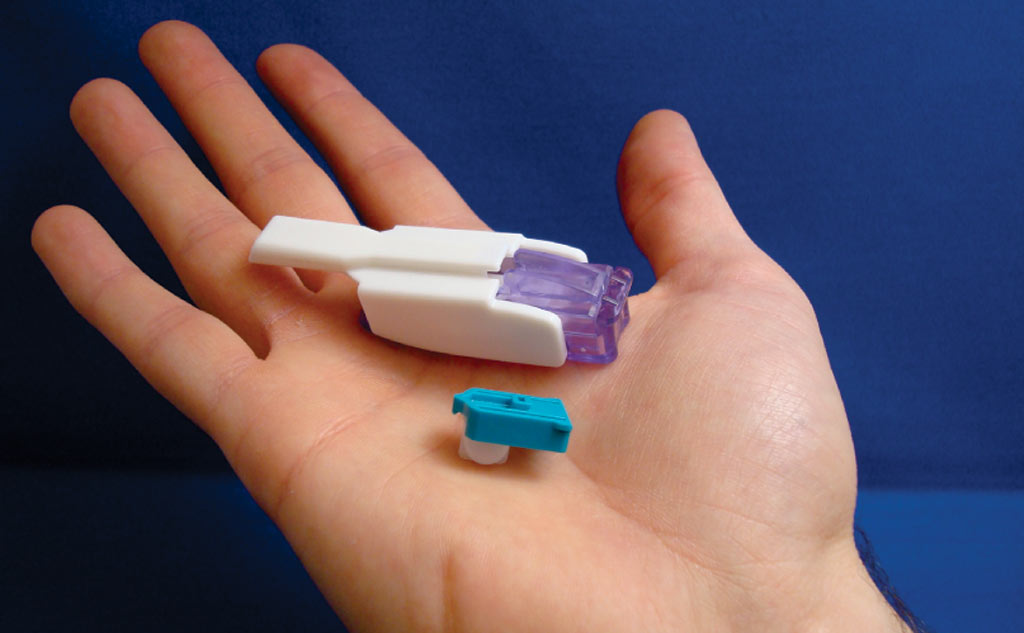
Afrezza is a powdered form of bolus insulin contained in an aspirin-size tablet and dispensed through a small inhaler, dubbed the Dreamboat, into the lungs. The technology developed by Mannkind allows only the insulin monomer molecule – the “active ingredient” in insulin – to bond to a fine, powdery substance that, when inhaled, quickly reaches the bloodstream through the lungs. In clinical trials with more than 6000 subjects, Afrezza’s insulin has interacted with glucose in a way that’s very similar to natural insulin. The insulin takes effect in 12 to 14 minutes (compared to 30 to 90 minutes for injected bolus insulin) and leaves the system within 3 hours (compared to 5 to 7 hours for injections).
The product appears to be a theoretical win-win for insulin-users. It delivers insulin in a manner that deals with “extra” glucose quickly, and significantly reduces the possibility of hypoglycemia due to that insulin’s rapid exit from the body. Afrezza could revolutionize insulin therapy, making it safer for people with Type 1 diabetes while vastly increasing its use and enabling better diabetes control among people with Type 2 diabetes. Founder Al Mann, who developed the insulin pump and the continuous glucose monitor, has spent more than a billion dollars of his considerable fortune pursuing the concept over the last 15 years. Afrezza’s clearance would be the capstone to Mann’s brilliant career as a technology and medical entrepreneur.
Report From The Front
The latest round of trials, undertaken after Mannkind received a FDA Complete Response Letter of the previous round of trials in 2011, tested Afrezza’s delivery method and effects among two groups of diabetics, one Type 1, the other Type 2. According to published reports, the Type 1 trials divided 518 Type 1s using basal-bolus insulin therapy into three groups: those using the newer Dreamboat inhaler, those using the first-generation Afrezza inhaler, and those using traditional insulin therapy by injecting subcutaneous insulin aspart (Novolog). Results showed no difference in pulmonary function between the new inhaler and the older inhaler, which will help Mannkind argue to regulators that the old pulmonary data should apply to the newer product. The data also showed that the Dreamboat-delivered insulin performed just as well as subcutaneous insulin aspart in decreasing A1C levels, and that the two groups using Afrezza suffered significantly fewer hypoglycemic episodes than their injecting counterparts.
If and when the product gets to market, reducing hypoglycemia potential among Type 1 users should be Afrezza’s greatest selling point among both endocrinologists and Type 1 patients.
The Type 2 trials compared people using Afrezza in combination with Metformin to Metformin users alone over a 24-week period, Afrezza-plus-Metformin users’ A1c levels dropped 0.82% during the trial, compared to a 0.42% decline in those using only Metformin, a clear win for Afrezza
The one negative to report was that people with Type 2 diabetes who were taking metformin and using Afrezza did experience more bouts of hypoglycemia than those using metformin alone, but researchers assumed this would occur because of the combined action of the two drugs on glucose levels. Episodes of severe hypoglycemia in patients with Type 2 remained statistically the same between those using Afrezza and those taking Metformin alone. This result perhaps underscores the value of the hypo-avoiding rapid exit effect observed among Type1 trial subjects. Therapeutically, Afrezza doesn’t overstay its welcome.
If all this sounds too good to be true – more effective and safer bolus insulin for Type 1 diabetes and an insulin alternative to shots for Type 2 diabetes – the bad news is that two previous submissions of New Drug Applications for Afrezza have been rejected by the FDA, including most recently in January 2011. Back then the Agency asked the company to conduct additional clinical trials, which are the ones Afrezza announced it just completed. Based on the data collected, Afrezza will take its third tilt at the FDA windmill sometime in October.
Once More Down the Home Stretch?
MannKind will submit its latest New Drug Application to the FDA in October, and wait for more questions or clearance. Given the higher threshold the FDA sets for clearing novel therapies and the rather mysterious on again-off again response in January 2011 to Afrezza’s 2010 New Drug Application, no one at the company will be holding their breath waiting for an answer. Still, with all the questions posed by the FDA in the 2011 Complete Response Letter apparently answered affirmatively by the latest studies, it’s hard for diabetes industry observers to see what the FDA could find wrong with these results.
If the FDA grants clearance, the final hurdle will be to bring the drug to market. MannKind has $10 billion worth of marketable inhalable insulin, purchased from Pfizer after that company withdrew its own insulin inhaler, Exubera, from market. Those stockpiles of insulin are stored in Germany, and a MannKind-owned plant in Danbury, Connecticut is ready to make the product and its packaging.
But mounting the kind of broad-based information and marketing campaign required to convince endocrinologists to prescribe Afrezza, let alone putting feet on the street to persuade primary care doctors, would be a massive undertaking. Diabetes industry analysts wonder whether one of the major players in insulin – Lilly, Sanofi, or Novo Nordisk – will acquire Mannkind or make a marketing and distribution deal with the company. No one knows, but such possibilities most certainly are being discussed now that MannKind is ready to take another swing at the fences of FDA approval.