A Type 1 Triple Play
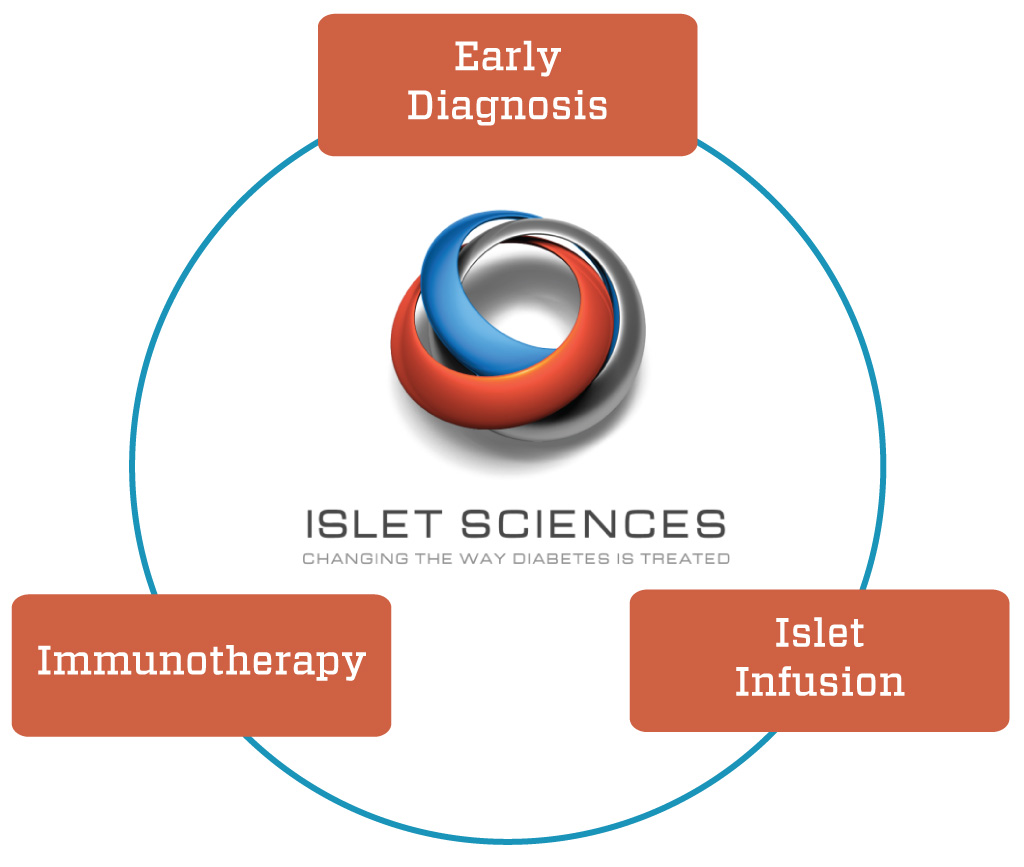
Most small companies targeting a specific disease hook their fortunes to a single product or therapy. Islet Sciences (www.isletsciences.com) takes a decidedly different approach. Founder and CEO John Steel, a Type 1 himself for 30 years, is betting that his young business can pull off a diabetes triple play, with products devoted to early detection, immunotherapy, and islet encapsulation and transplantation.
In essence, the outcomes he seeks are diagnosing Type 1 diabetes probability well before we can do it today, slowing its progress once diagnosed, and giving patients a do-over by injecting new islets, harvested in vast quantities from pathogen-free pigs, and protected from immune system destruction by a novel encapsulation process.
Success on any one of the three fronts would catapult Islet Sciences from promising startup to Wall Street darling, an outcome Steel has already made possible by listing the company’s shares on NASDAQ (Symbol: ISLT). Bringing one, any two, or all three products to market could be a gigantic success, both financially and for the people each would serve. Here’s a recap of each, presented in order of market size from largest to smallest.
“The Yale Technology:” Early Diabetes Diagnosis
The best way to prevent disease is to stop it before it starts, the public health basis of inoculation against once-common illnesses such as smallpox or polio. In the developed world, all agree that such preventive medicine is a good idea, as shown by the battery of routine shots given to every child who receives regular medical care. We don’t have a preventive shot for Type 1 diabetes yet, but we are developing ways to identify markers of a potential diabetes diagnosis years before the condition manifests itself. Islet Sciences is hot on that trail.
A year ago, the company exclusively licensed patented diagnostic DNA technology, developed by scientists at Yale University, which detects and measures beta cell death. We all know that beta cells are destroyed by the body’s own immune system in Type 1 diabetes. What if we could forecast which people would suffer from attack years before it happened? Islet Sciences believes the licensed Yale technology holds a key to forewarning, because it identifies circulating DNA derived from the cell-destroying process long before the actual destruction starts. Where there’s smoke, there’s fire, according to the theory.
The Yale technology might give clinicians a way to noninvasively detect and measure the health of anyone’s beta cells. “An early diagnostic test is important,” Steel says.
The Yale technology might give clinicians a way to noninvasively detect and measure the health of anyone’s beta cells. “An early diagnostic test is important,” Steel says. With this DNA test we can take a blood or urine sample and tell people if their islet cells are dying months or years before they are diagnosed with diabetes. We envision this test as something that everyone in the country, and possibly in the world, would get because everyone would want to know if their beta cells are working well or not,” says Steel. It isn’t hard to imagine the therapeutic and financial success of a diagnostic test given to billions of people. The hope is that this new technology could lead to earlier treatment, slowing the actual processes that cause diabetes.
This is, potentially, Islet Sciences’ “killer App.” In addition to signing an exclusive licensing agreement with Winthrop University Hospital for the Yale technology, the company is currently in discussions with pharmaceutical manufacturers to bring this test to market. A proven diagnostic product is two to three years away, in a best-case scenario.
LSF: Putting Diabetes In Slow Motion

While the diagnostic product might affect millions or even billions of people, the second step in Islet Sciences’ plan focuses on the millions of people in the U.S. currently using insulin therapy. To jump-start its entry into diabetes therapy, Islet Sciences acquired DiaKine Therapeutics, attracted by the biopharmaceutical company’s efforts to develop proprietary drugs that protect islet cells.
DiaKine’s most promising therapy is based on Lisofylline, or LSF, a synthetic molecule with novel anti-inflammatory properties. Stopping or reducing inflammation is important, because inflammation is what signals the immune system to send killer cells out to destroy insulin-secreting beta cells. LSF not only improves some aspects of cellular function, but also blocks other cells linked to beta cell destruction.
DiaKine and Islet Sciences already have an open Investigational New Drug application (IND) with the FDA for an injectable version of Lisofylline, and are in Phase II studies of an intravenously administered adjunct therapy to islet cell transplants.
Under its new ownership, DiaKine hopes to submit a New Drug Application to the FDA in 2014 for an orally administered version of Lisofylline to treat Type 1 diabetes, by stopping or slowing its destructive process. “We have data that show LSF is helpful to your heart and your nerves, and it rescues islet cells,” says Steel. Other compounds and drugs in DiaKine’s portfolio focus on beta cell protection. According to the company, LSF potentially could slow or halt the progress of newly-diagnosed Type 1 diabetes, treat or prevent insulin-dependent Type 2 diabetes, treat complications of diabetes such as peripheral neuropathy and retinopathy, and, of course, be an adjunct to islet transplantation.
End Game
Islet transplantation recipients, while small in number, are people with diabetes in dire need: they are at the stage where no conventional therapy helps them control the disease, and face certain death.
Of the three Islet Sciences products, Islet Science P™ will be the company’s first therapy to be tried in patients with diabetes. During the next year, it will be used in patients with end-stage kidney disease, a course of action recommended by the FDA. “We are going to the sickest diabetic patients and looking to rescue them first,” says Steel.
The two key areas impacting islet transplant availability, not to mention success, are the supply of islets, and the need for immune system suppression. Islet Sciences approaches the supply side through a relationship with the Spring Point Project, an FDA-approved facility for porcine tissue, so that the company can eventually supply all insulin-dependent patients with islet cell replacement therapy. The pigs raised by Spring Hope don’t live in a sty, but in a pathogen-free environment that allows harvesting of millions of islets that are very similar to those found in humans. Those old enough to remember that Banting and Best first secreted insulin by clamping the pancreas of dogs know that animal insulin has an honored place in diabetes therapy history. This is the newer, whiter, brighter version.
Islet Sciences P™ is encapsulated in a synthetic polymer using an alginate, or seaweed-derived “skin.” Alginates are widely used in many food products and the process is well understood.

Eventually, Steel believes, islet transplantation could be beneficial to all patients who are insulin-dependent. “If we can scale up what we do, this is something that everyone who takes insulin will want. It will make their disease management and clinical outcomes better. There’s strong evidence that if you can get normal glycemic control – without the ups and downs – you’re going to have a better health outcome.”
Islet Sciences sees its three technologies as being synergistic in the management of diabetes. “We see our technologies working this way: a person can have the DNA test, then use the drugs to slow down the diabetes, or get the islet replacement,” he says.
Islet Sciences has translated the research and developed or procured the technology. Now it’s working to get its technology and science into the hands of the clinicians who are diagnosing and treating patients. Will the triple play succeed? As all baseball fans know, they are rare, but completely possible. John Steel’s team is on the field, ready to pounce.