Sernova Cell Pouch Hopes to Improve Islet Transplant Success
Implant should increase long-term survival and function of insulin-producing cells

Sernova is a regenerative medicine company located in London, Ontario. The corporation is led by President and CEO Dr. Philip M. Toleikis (bio) who has a strong background in implantable medical devices from twenty years in leadership roles including Angiotech Pharmaceuticals, Inc.
We spoke with Dominic Gray, Corporate Communications, to learn more about Sernova’s current Phase I/II clinical trial. The Company expects to release initial safety and efficacy results throughout 2019.
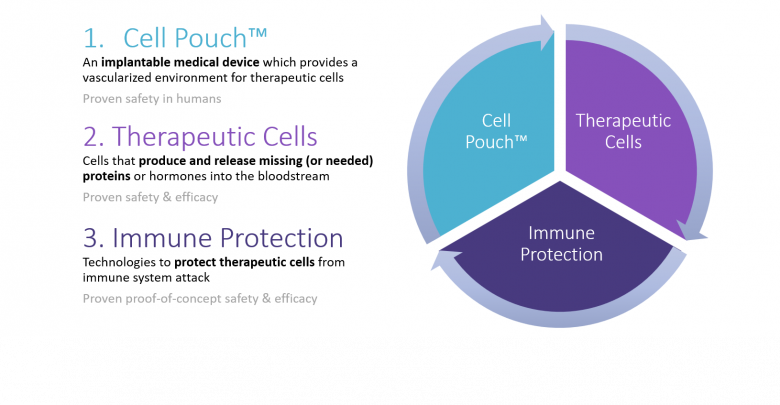
Cell Pouch
The Cell Pouch is a novel implantable and scalable medical device. It will create a long-term organ-like environment for islets and other cell therapies. The design of the therapeutic device aims to increase the survival and function of embedded therapeutic cells to better regenerate natural body functions, i.e., normal control of glucose levels in diabetes. Sernova’s Cell Pouch is specifically designed to incorporate with vascularized tissue and avoid fibrosis.
The Cell Pouch is inserted deep beneath the skin through a small incision in a minimally invasive outpatient procedure. This method allows for easy implantation and removal. Initially, the Cell Pouch implant will not contain any therapeutic content (i.e., no islet cells) to enable tissue chambers and blood vessels to develop within the pouch. Islets will then be transplanted into the Cell Pouch once vascularization occurs. The methods for this islet loading rely on Sernova’s patented transplantation loading device.
U.S. Clinical Trial at the University of Chicago
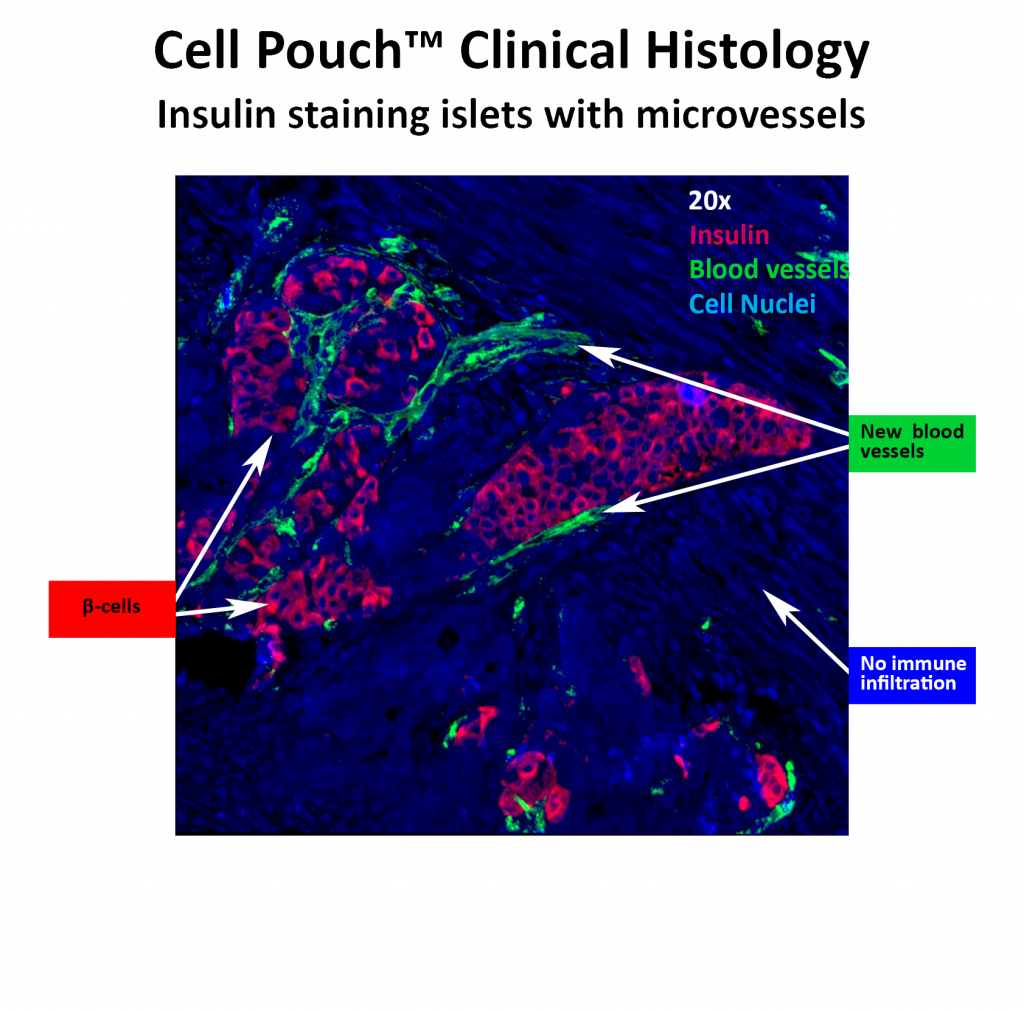
Sernova completed small and large animal safety and efficacy studies and conducted its first human trial in Canada. These studies showed that the Cell Pouch is safe, not rejected by the immune system and that islets transplanted within the device survive, become vascularized and produce the hormones required to control blood sugar levels.
Sernova then filed an IND (Investigational New Drug) application with the FDA and began screening patients in July 2018 for its new Phase I/II trial.
This new clinical trial is currently underway at the University of Chicago. Three patients out of the seven expected diabetic patients are already enrolled. A criterion for selecting a patient for the trial is that they have hypoglycemia unawareness. The clinical trial is to evaluate the safety and efficacy of the Cell Pouch in these patients.
Following the development of vascularized tissue chambers within the Cell Pouch, subjects will be stabilized on immunosuppression medications to protect the cells from an immune system response, and a dose of islets, under strict release criteria, will be transplanted into the Cell Pouch.
This current clinical trial is testing the safety and efficacy of the Cell Pouch System. This focus on the Cell Pouch itself is why this current trial is using systemic immunosuppression drugs to protect the islets from natural immune response.
The next clinical trial will test the safety and efficacy of local immune protection technologies currently in development. The objective of these new technologies is to adequately protect the islets from immune system attack and allow them to release their therapeutic factors without systemic immune suppression.
Following the first transplant. safety and efficacy will be measured for approximately 6 months for each subject. During this time, a small sentinel pouch also implanted will be removed after 90 days for an early assessment of the islet transplant. The degree of efficacy following an assessment at 6 months will help decide whether to transplant a second islet dose. The follow-up period after the final islet transplant is one year.
Initial results are expected to be released throughout 2019 and beyond, with final results released after the last patient follow up.
Unlimited Islet Cells: University Health Network Stem Cell Partnership
Islets are currently obtained from cadaver donors. This source is only sufficient to serve the most severe set of patients with hypoglycemia unawareness. There are not enough cadaver islets to treat all patients with diabetes.
To solve this limitation, Sernova has partnered with University Health Network (UHN) of Toronto for technologies related to differentiation of stem cells into glucose-responsive insulin-producing cells. The goal is to create an unlimited supply of ethically derived insulin-producing cells for treatment of diabetes within the Cell Pouch.
UHN insulin-producing cells will be tested in subsequent studies to confirm they have positive safety and efficacy.
Next Steps
Sernova aims to make its Cell Pouch System the new standard of care for several chronic diseases. The company is currently in active discussions to forge partnerships for the development, marketing and distribution of Sernova’s technologies for diabetes, hemophilia and other rare conditions.



![Researchers were able to preload enough insulin into the coin-sized adhesive microneedle patches to enable clinical use. [Image courtesy of UCLA]](https://insulinnation.com/wp-content/uploads/2020/03/Compressed-Patch-390x200.jpeg)



