Seraxis Islet Technology Aims for Practical T1D Cure
Seraxis has scalable islet production and a way to protect islets from rejection that avoids the need for immune suppression therapy
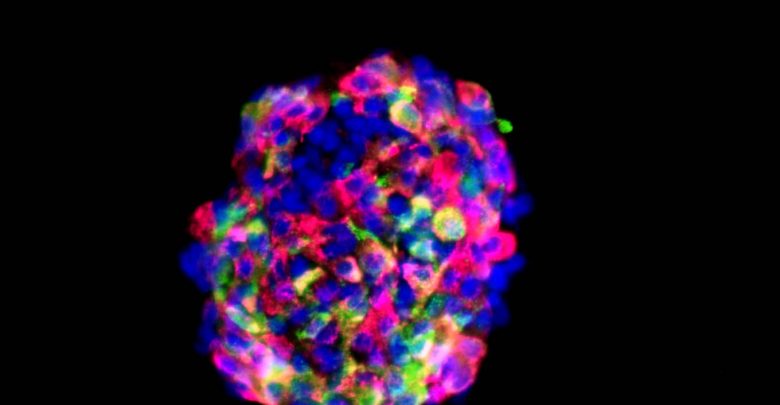
Islet transplants offer the hope of a permanent cure for Type 1 Diabetes. After nearly six years in stealth mode, Seraxis has emerged as a front-runner in the race to clinical islet deployment. Privately funded, and without any NIH or University affiliations, Seraxis has been remarkably efficient and comprehensive in their progress.
Islet Production
Seraxis has created a human islet factory that produces islets that closely mimic donor human pancreas islets. The Seraxis scientists started with donor human pancreas cells and converted these to induced pluripotent stem cells (iPSC). Unlike embryonic stem cells or other iPSC, these cells were specifically engineered to preferentially mature into their former identity: a mature pancreatic islet. Only a mature pancreatic islet can manage the complex, real-time regulation of blood glucose. The Seraxis maturation process takes about 30 days using a clinically compliant and scalable process. So far, the company has five granted patents for their islets and production process. These islets have a long shelf life and can be shipped worldwide in standard chilled containers already used for organ transplant shipments.
Islet Implantation
Consistent with the direction of work done at DRI (link — see the video in article), Seraxis places its islets on the omentum, a highly vascularized curtain of fatty tissue that seems to interact in complex and significant ways with the abdominal organs that make up our digestive system.
Unique to Seraxis is their method of protecting islets from rejection. SeraGraft is the name for a flexible thin macro-device consisting of a porous biocompatible polymer membrane that encapsulates a soft sponge-like matrix enriched with the Seraxis lab-grown islets. This device is roughly 4-inches by 5-inches (about the size of 3 credit cards) but only one-tenth of an inch thick.
The polymer membrane has been proven effective in other human applications and has tiny pores that are permeable to insulin, glucagon and other metabolites, but impermeable to cells. Thus, the body’s immune defenses can’t get in, and the islet clusters can’t get out.
Immune suppression therapy is not needed for recipients of cells within SeraGraft. The restrictive permeability of the outer layer does not permit entry by immune cells. The biocompatible materials of the device, coupled with its size, shape, and placement enable engraftment without stimulating a foreign body response and subsequent fibrosis.

Other islet transplant approaches place many free-floating microcapsules either under the skin or in the abdomen that, despite slick nonreactive surfaces, eventually have their effectiveness reduced by fibrosis as they settle and scar tissue surrounds each capsule. In contrast, the SeraGraft is designed to form a highly vascularized attachment to the omentum that avoids fibrotic accumulation.
“An important feature of the SeraGraft design is that it is scalable to accommodate any therapeutic dose in a single device. The SeraGraft can be removed and replaced, if necessary, ” said Seraxis CEO and Founder, Dr. William Rust.
Planned Clinical Procedure
Installation of the SeraGraft is planned as an outpatient laparoscopic procedure. The SeraGraft is inserted as a tightly rolled tube via a one-inch incision and then unrolled onto the placement site, for anchoring to the omentum.
Self-Regulating Therapy
The islet clusters in the SeraGraft produce the usual mix of glucagon, insulin and other hormones. It works effectively as a surrogate pancreas. During in vivo testing in diabetic animals, the SeraGraft never overdosed the animals, just like a healthy pancreas does not lead to hyper- or hypoglycemia.
Clinical Trials
The Seraxis team is working on its clinical trial application and is applying Good Manufacturing Practices to their islet production. Seraxis has completed a Pre-IND (Investigational New Drug) Consultation with the FDA and is hiring staff and building out facilities in anticipation of an IND approval in the near term.
Funding its clinical trials is the next step that Seraxis plans. After that, it is uncharted territory.
Seraxis anticipates a particular clinical trial designated phase 1/2, to address both safety and efficacy. If that produces the anticipated results in adult patients, the Seraxis T1D islet therapy could pursue an accelerated path through clinical approval. The Seraxis team is also exploring clinical trials in South Korea, Singapore or Japan; locations, where cell therapy regulations are streamlined compared to those in the United States.
Season of Hope
A T1D cure is a perennial aspiration for us all. Good treatment approaches and a positive attitude produce good results, but a cure would be fantastic. In this season of hope, we particularly wish Seraxis good luck!
Islet Questions and Answers
What are islet cells?
Pancreatic islets, also called islets of Langerhans, are groups of cells in your pancreas. The pancreas is an organ that makes hormones to help your body break down and use food. There are about 1 million islets in the pancreas, and Islets contain several types of cells, including beta cells that make the hormone insulin. (NIH)
Who discovered them and when?
Paul Langerhans (25 July 1847 – 20 July 1888) was a German pathologist, physiologist, and biologist, credited with the discovery of the cells that secrete insulin, named after him as the islets of Langerhans. (Wikipedia)
What is an Islet Transplant?
The goal of an islet transplant is to augment the recipient’s body with healthy islet clusters from a donor source, generally cadavers. The primary transplant method injects these harvested donor islets into the recipient’s liver. The liver transplant regimen includes two to four injections of 500,000 islets each along with heavy doses of immune-suppression drugs. The recipient’s body will still trigger an immune response that will kill a large portion of the new islets. The surviving islets produce insulin, glucagon, and other metabolic hormones to aid use and control of glucose.
Why haven’t islet transplants been used to solve diabetes yet?
The procedure to transplant islets is costly. The sources of donor islets are limited. It can take up to two years to find a compatible donor. The long-term use of immune-suppression drugs has damaging side effects, including bacterial and viral infections, sores, diarrhea, high cholesterol, high blood pressure, fatigue, and even some cancers.
Does an islet transplant mean no more insulin?
A majority of islet transplant recipients do not require supplemental insulin for a while, with a minority not requiring supplemental insulin five years after the transplant.
How have islet transplants evolved?
Research into islet transplants has focused on finding new sources of donor islets, often starting with stem cells. Seraxis is taking the promising approach of developing a cell therapy from induced pluripotent stem cells, unlike other existing efforts which focus on embryonic stem cells.
New encapsulation approaches have been developed to protect the islets from the body’s immune system including both micro-encapsulations of individual islet clusters and macro-encapsulation to safeguard the entire dose of islets. DRI’s BioHub and the Seraxis SeraGraft are examples of macro-encapsulation.
The liver was the original site for placing the donor islets. New sites are being explored, including subcutaneous tissue and the omentum.
How do I qualify for an islet transplant?
The FDA classifies islet transplants as experimental, and they are only approved for adult patients who are part of a research study. A patient must have a history of severe hypoglycemia and hypoglycemia unawareness. The procedure is only approved for people over 18. It is not approved for people with type 2 diabetes. In total, about 1,000 islet transplants have been done in the United States so far.
Would islet cell transplants work for type 3C diabetes (pancreatogenic diabetes)?
Theoretically, yes but this does not seem to be the focus of current research.
Why not just transplant a whole pancreas?
Some T1D people do receive a whole pancreas transplant, usually along with new kidneys in people with kidney failure. According to the National Kidney Foundation, more than 5,000 pancreas transplants have been performed in the U.S. since 1968; 95% are in combination with a kidney transplant. After five years, about 42% are still functioning well. “Whole-organ pancreas transplants are fraught with complications,” says F. Charles Brunicardi, M.D., F.A.C.S., Professor and Vice Chair of Surgical Services at the David Geffen School of Medicine at UCLA and Chief of General Surgery at, Santa Monica Hospital. “It’s better to leave a recipient’s pancreas alone—it still functions perfectly well, producing enzymes needed to digest food—and transplant the islet cells elsewhere.”
Sources
We interviewed Seraxis executives Carole A. Welsch, Ph.D., M.B.A., Chief Business Officer, and William Rust, Ph.D., CEO. See http://www.seraxis.com/
Recent press releases from Seraxis:
http://seraxis.com/wp-content/uploads/2017/03/Seraxis-Press-Release_Sept-5-2018.pdf
Previous articles in Insulin Nation on islet replacement:
https://insulinnation.com//?s=islet
Diabetes Research Institute Clinical Trial Testing the Omentum: https://www.diabetesresearch.org/islets-in-omentum/







