Pramlintide Reimagined and Results are Promising

Over a decade ago, the US approval and launch of pramlintide (Symlin® AstraZeneca) brought a lot of excitement because of its therapeutic ability, when used in combination with mealtime insulin, to flatten post-meal blood glucose levels, reduce glucose excursions, and cause weight loss – and pramlintide accomplishes all this with about 20% less insulin use.
Studies have shown that pramlintide accomplishes this by increasing the feeling of fullness (thus reducing food intake), delaying the rate at which food moves from the stomach to the intestine, and reducing endogenous glucose from the liver by suppressing glucagon production.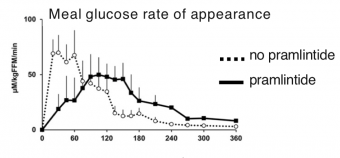
Pramlintide is an injectable amylin analog drug for both Type 1 and 2 diabetes and is the only FDA-approved drug to lower blood sugar in Type 1 diabetics since insulin’s approval in the 1920s. In normal physiology, amylin is a hormone that is co-secreted into the bloodstream at a fixed ratio with insulin by the β cells of the pancreas.
The existing product label for pramlintide warns of severe hypoglycemia, which may have resulted from poor design of the initial phase 3 trials that did not modify insulin doses despite the addition of an additional glucose-lowering agent. Nausea with pramlintide was a real challenge for many users, especially in T1D, as people would bolus their insulin – along with a separate injection of pramlintide – for an intended meal, become nauseated by the pramlintide, and subsequently not eat the original amount of food. The resultant mismatch between food intake and insulin dose could lead to hypoglycemia or an increased risk for hypoglycemia. Careful titration of both insulin and pramlintide, especially in T1D, is needed for success with pramlintide.
With pramlintide’s indication for use at all major meals, people already on the 4 shots of a basal-bolus insulin regimen faced the addition of 3 or more shots per day, a challenging proposition. Additionally, the daily pramlintide shot burden made it unattractive to pump users who often chose pumps to avoid shots. For all of these reasons, current use of pramlintide is quite limited as the burdens (shots, hypoglycemia risk, nausea, cost) outweigh the perceived benefits. These likely play into the fact that there is very little promotional effort to the product today.
Pramlintide, because of it flattening action on glucose (lowering highs and reducing lows) also has limited impact on A1C and, in the past, this suggested modest efficacy in a market focused on A1C as the best measure of control. With increasing use of CGM, however, the main benefit of pramlintide, i.e. glucose flattening, is far more recognized and appreciated.
Pramlintide Reimagined
Adocia, a biotechnology company located in Lyon, France, is utilizing its BioChaperone (BC) platform to develop innovative formulations of therapeutic proteins. The company has 34 issued patents around the BioChaperone family of excipients. Adocia’s innovative BioChaperone technology serves as a vehicle to enhance a drug’s stability and solubility, a key feature for efficient diabetes drug delivery – and especially useful in multi-drug combinations which are emerging as a new approach in diabetes treatment.
Adocia is applying its BioChaperone platform to develop a single injectable solution that combines insulin and pramlintide in a fixed ratio. The combination of these two agents is challenging to achieve because pramlintide and prandial insulins are naturally separate, difficult to mix and form precipitates when combined.
However, Adocia’s BioChaperone Pram Insulin has remarkable properties. It is physically stable with a two-year shelf life when refrigerated and 30 days after that at up to 30℃, with chemical stability comparable to insulin in vials and pumps.
Adocia’s BioChaperone platform enables the novel combination of the two agents, insulin and pramlintide, for BC Pram Insulin. The titratability of the combination, as compared to fixed dosing for pramlintide alone, may help to overcome some of the issues (e.g., nausea, hypoglycemia) of the separate administration of the two agents.
In preclinical studies, BC Pram Insulin has demonstrated extended exposure and duration of glucose flattening compared to separate injections of insulin and pramlintide.
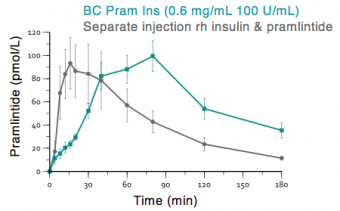 In early September 2018, Adocia announced positive topline results for the first clinical study of BioChaperone® Pramlintide Insulin in people with type 1 diabetes (release).
In early September 2018, Adocia announced positive topline results for the first clinical study of BioChaperone® Pramlintide Insulin in people with type 1 diabetes (release).
The topline data from the first in human trial showed a 97% reduction in 2 hour PPG AUC versus Humalog.
Additional data on the BC pramlintide program is expected to be released at a major upcoming conference.
The company selected the ratio being evaluated in clinical studies based on research work conducted by Dr. Matt Riddle and the JDRF indicating a target range of pram/insulin ratios with superior therapeutic value.
Dr. Matthew Riddle, Professor of Medicine, Oregon Health & Science University, Division of Endocrinology, Diabetes, & Clinical Nutrition, previously commented, “These are encouraging results, confirming that a co-formulation of pramlintide with human insulin can reproduce the marked reduction of postprandial glucose shown in previous studies of pramlintide and insulin given separately. By mimicking the normal co-secretion of amylin and insulin with meals, this 2-in-1 combination has the potential to address the persisting unmet need for mealtime glucose control for people with diabetes.”
It is interesting to note that Adocia uses human insulin in their BC pramlintide as the slowed post-meal glucose appearance better matches the insulin action profile of human insulin, eliminating the need for the speed of an insulin analog.
 Adocia is focusing on combining pramlintide and insulin – working on multiple combinations – as they envision this product as a potential revolution in glucose management.
Adocia is focusing on combining pramlintide and insulin – working on multiple combinations – as they envision this product as a potential revolution in glucose management.
The company has another phase 1b trial scheduled to start in early 2019.
We are encouraged by Adocia’s approach and will continue to watch the biotech’s advancing pipeline progress. In connection with the growing use of CGM, the glucose flattening benefit of pramlintide combined with Adocia’s unique chaperoning drug delivery technology could potentially be a real win for everyone using insulin therapy for their diabetes treatment plan.







