CGM Sensors are good and getting even better
Diabetes technology has improved rapidly and advances in Continuous Glucose Monitors, Insulin Delivery, and Artificial Pancreas are coming
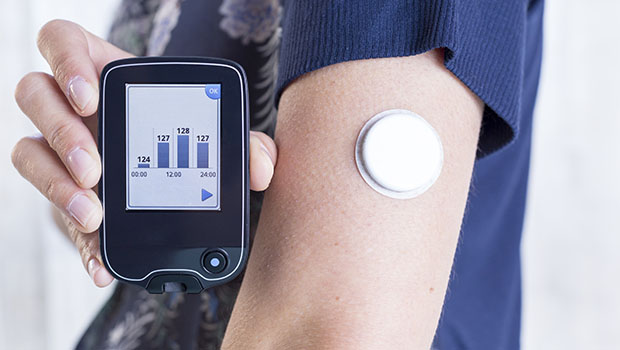
MediSense, a UK/US company acquired by Abbott in 1996, was the first to offer a biosensor for BG monitoring. Thirty years later, the Abbott FreeStyle Libre system is the current product of that team’s ongoing efforts.
We spoke with John Willis, Ph.D., CEO of Ultradian Diagnostics LLC (bio) who worked at MediSense and Tom Bishop (bio), who was the VP of Product Development at Echo Therapeutics and actively consults to medical device firms, to get their thoughts on the evolution of CGMs based on their long careers in BG monitoring and medical devices.
The vast majority of CGMs use sensors based on enzymes to detect glucose. This technology is widely understood has been used commercially for over 30 years. Recently new sensor technology has come out of the labs and has been approved by the FDA. So at a high level, there are two major sensor approaches, and both achieve excellent results.
CGM Sensor Options Today
Enzyme-based sensors are used by Medtronic, Dexcom and Abbott FreeStyle and typically have a 7- to 10-day useful life.
Science: The enzyme glucose oxidase reacts with glucose, water, and oxygen to form gluconic acid and hydrogen peroxide. The hydrogen peroxide is detected with a simple electrical circuit. The more glucose in the blood, the more current that flows thru the circuit.
Medtronic and Dexcom make sensors that continuously measure the glucose and thus can produce alarms when a patient has become hypoglycemic (low) or hyperglycemic (high). The Abbott system is called a “flash” glucose monitoring system; it reports glucose values only when the patient places a reader near the sensor
A fluorescent polymer approach is used by Senseonics in their Eversense embedded sensor product. In the US these sensors have a 90-day life while in Europe they are labeled for up to 180-days with a 365-day model in trials.
Science: The Senseonics system has three parts: a sensor that is surgically implanted under the skin, a transmitter worn in an armband and a Smartphone app. The patient’s glucose diffuses into the sensor. The receiver then illuminates a fluorescent sensor that detects the glucose.
CGM Sensors will Improve
There are pressures to further enhance CGM technology:
- Factory calibration is gaining momentum, with both the Dexcom G6 and the Abbott Freestyle allowing use without finger-sticks.
- Increase mean accuracy will improve from current ISO requirements of 99% of results within the A (±20%) & B (±30%) Zones of the Consensus Error Grid.
- Adhesives that accompany sensors will improve to last 10 days or more.
- Reduced costs. Abbott has turned the CGM world upside down with its lower cost sensor. This has begun a competitive war that will benefit people with diabetes worldwide by pushing down the prices paid for sensors.
“About 80% of revenue for CGM and Pump suppliers comes from Europe and North America. Despite huge populations of prospective users, the rest of the world is not a major market because of the high costs of the devices and consumables.
Today, most minimally invasive sensors cost $60 to $70 each. Online sources may save 20%, but the annual cost of sensors is at least $2000. There is pressure to increase the useful life of sensors because people often use sensors beyond their design life and then obtain unreliable readings. This pressure to increase useful (design) life while also improving accuracy to meet ISO demands is likely to keep sensor prices high for years.
Black Swan Disruption
There are black swans on the horizon for CGMs in the form of non-invasive or minimally-invasive CGM solutions. Apple, for example, has been working on an optical sensor for use with the Apple Watch for many years. Other companies, such as PK Vitality in France are bringing out CGM sensors that use novel sensing technology.
Perhaps anticipating a disruptive entry we may see future systems that integrate both the sensor and pump into the single disposable wireless package. This multifunction package would justify continued high prices for each disposable.”
Stay Tuned
Things are changing very quickly in the world of Diabetes equipment. Tom Bishop has agreed to write three articles for us:
- Advanced CGM Systems and Technologies
- Advanced Insulin Delivery and Management
- Artificial Pancreas – Will it really work?







