Will ViaCyte’s Encaptra Cell Delivery System ‘Cure’ Type 1 Diabetes?
Here is an update on ViaCyte’s approach and progress to advance its scalable islet transplantation system through the FDA approval process
When you were diagnosed with type 1 diabetes, you were probably told that a cure was “just around the corner.” And that “corner” likely came and went years or decades ago.
While a true cure isn’t exactly just a few years away, there are several remarkable efforts towards eliminating the need for ‘exogenous’ insulin in people with type 1 diabetes.
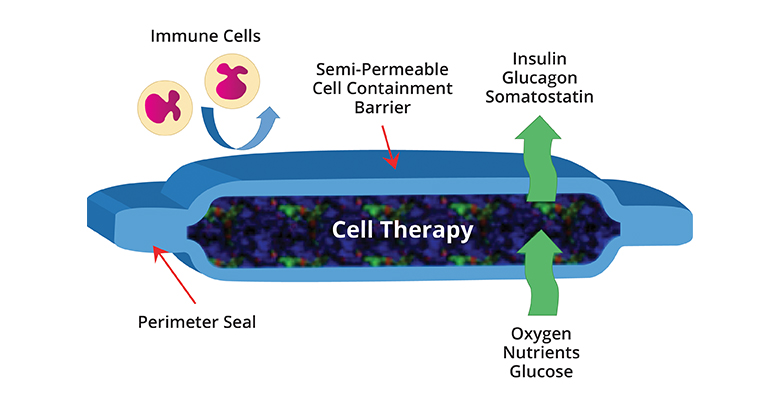
One of the most notable efforts is ViaCyte’s Encaptra “cell delivery system.” This is different from the islet transplantation you have read about where keeping the islets healthy is difficult. Instead, ViaCyte transplants pancreatic beta-cell ‘precursors’ that mature in place as needed into islets.
What needs to be ‘fixed’ in your body to be ‘cured’?
To understand a ‘cure’ for type 1 diabetes, it’s important to first understand what beta-cells are. While we often blame our pancreas for not doing its job of producing insulin, the real issue is that your pancreas tries to produce insulin every day but your immune system continues to attack and destroy the beta-cells secreted by your pancreas that also produce insulin.
If you simply received a pancreas transplant, your immune system would just attack and destroy those beta-cells, too, without tremendous immunosuppression drugs that come with a slurry of other issues and truly isn’t a practical or safe approach for the average patient.
This means that today’s approaches towards a cure need to either reprogram your immune system to specifically stop attacking your beta-cells or you need beta-cells introduced in a way that protects them from your immune system altogether.
That’s where ViaCyte’s research comes in.
What is ViaCyte’s Encaptra ‘cell delivery system’?
ViaCyte — based in San Diego, CA — has developed a device that is implanted in a person with Type 1 Diabetes. There are actually two devices — PEC-Encap and PEC-Direct — that work very similarly. Both contain pancreatic beta-cell ‘precursors.’
Beta-cell precursor cells are basically like the newborn baby of a fully functioning beta-cell. These young beta-cells develop into mature islet cells that are able to produce insulin, amylin, and other endocrine hormones.
ViaCyte’s technology is one of the leading approaches to ‘curing’ type 1 diabetes and has proven effective in over 1000 rodents with type 1 diabetes along with many ongoing human patient trials.
Let’s take a closer look.
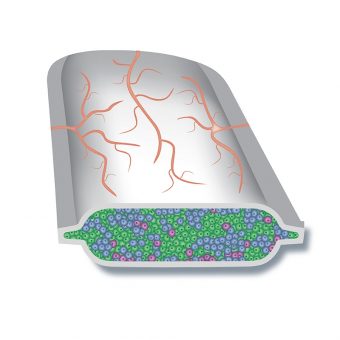
The PEC-Encap
This is a device that is completely encapsulated which means it doesn’t require suppressing your immune system. Instead, it contains beta-cell precursors that are fully protected from being attacked and destroyed.
The device is implanted within the subcutaneous layer of your skin — which means its fortunately very small, the process of implanting it is minimally invasive, and the recovery period is very short. Over the course of several months, the surface of the device becomes gradually ‘vascularized’ which means that glucose, oxygen and other nutrients can pass into the device and insulin and other hormones can pass back into the blood vessels on the device surface.
This vascularizing triggers those beta-cell precursor cells to mature into insulin-producing cells. So, even though your immune system cannot breach the device and destroy the cells, your blood glucose can flow through it and insulin and amylin can be returned to the rest of your body.
Of course, the supply of beta-cell precursors isn’t endless. At this point, ViaCyte’s device requires a routine implant every year to provide more cells. Overtime, ViaCyte’s goal is to extend the time between implants.
Another overarching goal is to make the implant process itself faster, easier, and less invasive, without any additional cell implants at all.
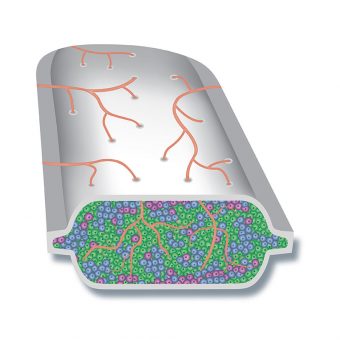
The PEC-Direct
Very similar to the PEC-Encap, PED-Direct technology from ViaCyte is intended only for those with higher-risk type 1 diabetes, like patients with severe hypoglycemia unawareness and consistently unpredictable swings in blood sugar levels, often referred to as “brittle diabetes.”
This device enables blood to flow into it and intermingle with the contained cells rather than remaining on the surface. Thus, this technology requires a constant suppression of the immune system.
- Suppressing your immune system to protect beta-cells inevitably means also suppressing your immune system’s ability to protect you from other things like the common cold and the flu.
- We underestimate just how much our immune system does for us on a daily basis until it’s suddenly been put on vacation and we become very vulnerable.
- It simply isn’t ideal to suppress the immune system without a life or death reason to do so.
PEC-Direct is being tested for those people whose situation requires it.
ViaCyte’s FDA Journey
When can I sign-up for a ViaCyte implant?
You can’t rush awesome things — including the cure for type 1 diabetes. ViaCyte’s cure technology has come a very long way in the required research process but it still has a ways to go.
How long until ViaCyte’s cure treatment is widely available?
There are 4 phases any medical technology must go through before it becomes approved by the Food & Drug Administration and available to the general public. Fortunately, ViaCyte is already halfway through this journey.
- Phase I: Safety & Tolerability
- Phase II: Efficacy & Side-Effects
- Phase III: Efficacy, Superiority & Monitoring of Adverse Reactions
- Phase IV: Submit for FDA approval
Phase II is Still Recruiting
Begun in July 2017 with an estimated end in September 2020 ViaCyte’s Phase II trial is still recruiting additional patients for this research. This is an exciting phase where the technology is demonstrated on humans instead of rodents.
Participants must meet all of these details in order to qualify:
- Men and non-pregnant women of non-childbearing potential
- Diagnosis of T1DM for a minimum of five (5) years
- At least one (1) severe hypoglycemic event in the previous 12 months
- Hypoglycemia unawareness or significant glycemic lability
- Stable diabetic treatment
- Willingness to use a continuous glucose meter
- Acceptable candidate for implantation
Patients with any of the following issues cannot participate in the study (however, that doesn’t mean they wouldn’t be able to receive the technology once it’s FDA-approved!):
- History of islet cell, kidney, and/or pancreas transplant
- Six (6) or more severe, unexplained hypoglycemic events within six (6) months of enrollment
- Uncontrolled or untreated thyroid disease or adrenal insufficiency
- Diabetic complications such as severe kidney disease or renal dysfunction, proliferative retinopathy, diabetic foot ulcers, amputations attributable to diabetes, and/or severe peripheral neuropathy
- Non-compliance with the patient’s current anti-diabetic regimen
Overall Timing
It’s exciting and stressful all at the same time because naturally, we want it to be available right now. It’s possible that we may see FDA-approval for ViaCyte’s technology in 10 years, around 2030. In the meantime, please keep taking your insulin and checking the ol’ blood sugar!







