The Cure is in Us

11-year-old Louis Cocco’s head was barely visible above the podium as he addressed an attentive group in a conference room at the Cannon House Office Building in Washington, D.C. this past April. Louis, who has Type 1 diabetes, rattled off the statistics about the health and financial impacts of the disease like a public-speaking veteran. Then, at the end of his talk, he turned towards Dr. Claresa Levetan, his endocrinologist and the organizer of the meeting, and said, “I know this group of doctors will get me off insulin before I am a grownup.”
If you are unfamiliar with Levetan’s track record, you might condemn her for offering Louis false hope, but she is not some quack selling diabetes snake oil. Levetan is a Fellow With Distinction of the American College of Endocrinology, whose pioneering work has received many accolades. She has received the prestigious Roche Diagnosed award for innovative diabetes research and has been an invited visiting scholar at Harvard University’s Joslin Diabetes Center. Dr. Irl Hirsch, editor-in-chief of the ADA journal Clinical Diabetes has called her “perhaps the most creative physician I have ever met.”
Her latest endeavor is a company called Perle Bioscience www.PerleBioscience.com, and her promise to Louis is founded on developments in its labs. Levetan hypothesizes that a cure for diabetes, Type 1 or Type 2, can be achieved by our ability to regenerate islets inside ourselves, using our own tissue, and combining regeneration with immune suppression. She sees diabetes remission as a 2-part, intertwined process wherein the body grows healthy new islets and also suppresses immune system attacks. A combination therapy like this has not been attempted in treating diabetes, but it will be soon. Human trials will begin in both the United States and Europe later this year.
Been There, Done That
If the past is prologue, Perle’s recent discoveries and proposed therapies might make Levetan and her backers another tidy fortune, in addition to making diabetes go away. Her first company, CureDM, licensed another regenerative islet cell therapy, called Human proIslet Peptide (HIP/Pancreate), to Sanofi in 2010, in a deal worth $335 million if and when a finished product comes to market. Sanofi is quiet about what it is doing with HIP, but one thing they have made clear to Levetan is that they are focusing their development and testing on Type 2 diabetes. She, on the other hand, is committed to helping Type 1s. “Our mission is to make people with Type 1 diabetes insulin independent,” she says, “and it never stops.”
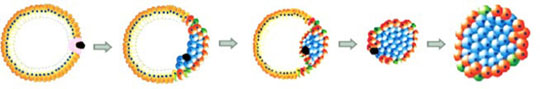
Levetan’s research, at CureDM and today, has identified a specific human regulatory gene, known as REG, which is usually found only in fetal development, when the human pancreatic islets are first formed. REG doesn’t disappear after birth; it lies dormant, waiting for something to create a need for its services. It can reappear when there is acute pancreatic injury, and has been shown in other studies to transform human pancreatic ductal tissue into new beta cells. The HIP therapy licensed to Sanofi is based on this concept.
Pancreatic regeneration from ductal tissue is not just Levetan’s theory. Researchers at Yale and Harvard have confirmed the presence of the REG gene protein at the time of new onset Type 1 diabetes, and in the very newest islets formed from pancreatic ducts. Scientists at Novo Nordisk have shown that the HIP directly forms new islets from pancreatic ductal tissue.
Levetan barely paused her work after CureDM gave up control of HIP’s development 3 years ago. She and her team have identified 4 new REG gene peptides that bind directly to the pancreatic receptor in ductal tissue. The acronym for these peptides is BRAD, short for Beta Regeneration Agents for Diabetes. When BRAD binds to the ductal tissue receptors, it starts a process that produces new beta cells. Perle Bioscience has filed four “composition of matter” patents to protect its discoveries. In all, the company has nine diabetes intellectual property patents pending.
Back To The Future
Levetan’s regeneration concepts are not new. More than a century ago, scientists believed that the pancreas contained its own islet regeneration system that would respond to severe pancreatic injury by creating new cells. Frederick Banting, a pioneer in the discovery of insulin, subscribed to the idea. In Banting’s time a little under a hundred years ago and before insulin was widely available, surgeons would perform partial pancreatectomies and tie off the pancreatic ducts in diabetic children to stimulate the growth of new cells. The procedure worked, but diabetes remission seldom lasted more than two years, which we now can attribute to repeated immune system destruction of the new beta cells. Diabetes wasn’t recognized as an autoimmune disease until the 1980s, and no one in Banting‘s time studied either the possibility of using a therapy like HIP to regenerate one’s own islets or methods to suppress the human immune system to combat diabetes.
Another thing Banting and his peers could not have known was the complex relationships between beta cells and the other cells in human islets. While insulin-producing beta cells are the most numerous occupants of an islet, other cells (alpha, delta, gamma and epsilon) all play a role in metabolic function and glucose conversion by secreting amylin, glucagon, somatostatin, ghrelin and pancreatic polypeptide. The cells communicate with each other and the brain, and depend on each other for normal glucose metabolism, which maintains 95% of normal glucose levels below 120 in patients without diabetes. When the immune system attacks and kills the beta cells, the other cells stop functioning, too. Regenerating islets from your own tissue, rather than transplanting islets from a cadaver, will be more effective. Or so the theory goes.
Cut To The Chase: Human Trials First

The theory is about to be tested in people, not mice. Perle’s ability to road-test Levetan’s ideas in human subjects is based on the design of its first 2 trials, which will use 2 existing drugs, cyclosporine and lansoprazole, to show proof of concept. Sometime before the end of 2013, 120 subjects in 2 groups of 60 will be enrolled in 2 year-long trials. One group will consist of newly diagnosed Type 1s. The other will be people who have had Type 1 for years. Some in each group will be given doses of the 2 existing oral drugs, twice a day, while others will receive placebo therapy. Since both cyclosporine and lansoprazole have well-established safety track records in thousands of patients with Type 1 diabetes, there should be few, if any, side effect surprises.
Cyclosporine is an immunosuppressive drug regularly prescribed to prevent immune system rejection in people who have undergone heart, liver, and kidney transplants. In the 1980s and early 1990s, it proved able to cause diabetes remission within 7 weeks of initiation in 2/3 of Type 1 patients who started using it right after diagnosis. Remission usually lasted two years or less, however, and the therapy was abandoned for diabetes because it didn’t produce a long-term cure. At the time, of course, there were no regeneration agents available.
Lansoprazole in the U.S. is found in an over-the-counter medication called Prevacid24HR, marketed by Novartis. The drug helps to increase levels of gastrin in the body while combating various issues like gastric reflux. Way back in 1955, it was also shown to potentially increase beta cell formation as a function of increased gastrin production.
While the first 2 trials are running, Perle will invest in further research on the BRAD peptides and hopes to be able to request a “fast track” status from the FDA for an Investigational New Drug (IND) approval, using the new drugs along with cyclosporine. Fast track status for a Perle IND would be based on the combination therapy’s theoretical ability to reverse diabetes at onset among the 50,000 children who are diagnosed as Type 1s each year in the U.S. BRAD’s future use, of course, depends to some extent on the results of the preclinical BRAD studies and the human trials of cyclosporine and lansoprazole, which will test the hypothesis that both an immune agent and a regeneration agent are required for insulin independence.
Levetan, a soft-spoken Southerner whose accent belies the location of her private endocrinology practice in suburban Philadelphia, is pleasant but determined when talking about her work. “I promised Louis a cure,” she says. “I intend to make that promise come true.”
Let’s hope that she can.